Resources
Events
 The Lodge – Colonial Williamsburg, VAApril 24-26, 2026CHES FoundationLadyBugs National Women’s Program: Women with Bleeding Disorders, Caregivers, Carriers
The Lodge – Colonial Williamsburg, VAApril 24-26, 2026CHES FoundationLadyBugs National Women’s Program: Women with Bleeding Disorders, Caregivers, Carriers Minneapolis, MNApril 29-May 2, 20262026 ASPHO ConferenceAdvancing Knowledge, Discovery and Collaboration
Minneapolis, MNApril 29-May 2, 20262026 ASPHO ConferenceAdvancing Knowledge, Discovery and Collaboration Paris, FranceJuly 11-15, 2026ISTH 2026 CongressInternational Society on Thrombosis and Haemostasis
Paris, FranceJuly 11-15, 2026ISTH 2026 CongressInternational Society on Thrombosis and Haemostasis Orlando, FloridaAugust 13-15, 2026Bleeding Disorders Conference 2026National Bleeding Disorders Foundation
Orlando, FloridaAugust 13-15, 2026Bleeding Disorders Conference 2026National Bleeding Disorders Foundation The Westin Detroit Metropolitan AirportOctober 2-4, 2026HFM 2026National Conference for Women with Hemophilia and Rare Factor Deficiencies
The Westin Detroit Metropolitan AirportOctober 2-4, 2026HFM 2026National Conference for Women with Hemophilia and Rare Factor Deficiencies Hilton Boston Park Plaza, Boston, MAOctober 9-11, 2026FWGBD 2026The Foundation for Women & Girls+ with Blood Disorders 4th National Biennial Conference
Hilton Boston Park Plaza, Boston, MAOctober 9-11, 2026FWGBD 2026The Foundation for Women & Girls+ with Blood Disorders 4th National Biennial Conference InterContinental New OrleansOctober 21-22, 2026ATHN 2026ATHN Data Summit
InterContinental New OrleansOctober 21-22, 2026ATHN 2026ATHN Data Summit Tampa Marriott AirportNovember 13-15, 2026CHES FoundationOne Drop Rare Bleeding Disorders Consortium
Tampa Marriott AirportNovember 13-15, 2026CHES FoundationOne Drop Rare Bleeding Disorders Consortium
Publications


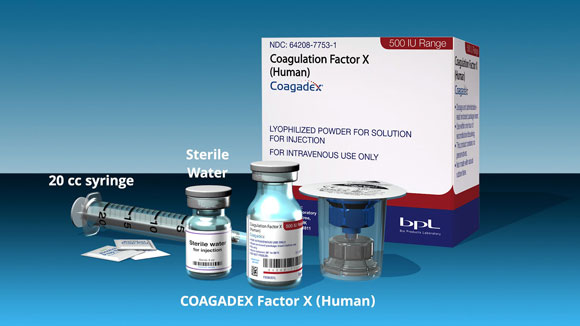
Indications and Usage for COAGADEX
COAGADEX, a plasma-derived blood coagulation factor X concentrate, is indicated in adults and children with hereditary factor X deficiency for:
- Routine prophylaxis to reduce the frequency of bleeding episodes
- On-demand treatment and control of bleeding episodes
- Perioperative management of bleeding in patients with mild, moderate and severe hereditary factor X deficiency
Contraindication for COAGADEX
COAGADEX is contraindicated in patients who have had life-threatening hypersensitivity reactions to COAGADEX.
Important Safety Information for COAGADEX
Allergic type hypersensitivity reactions, including anaphylaxis, are possible with COAGADEX. If symptoms occur, patients should discontinue use of the product immediately, contact their physician, and administer appropriate treatment.
The formation of neutralizing antibodies (inhibitors) to factor X is a possible complication in the management of individuals with factor X deficiency. Carefully monitor patients taking COAGADEX for the development of inhibitors by appropriate clinical observations and laboratory tests.
COAGADEX is made from human plasma and may contain infectious agents, e.g. viruses, the variant Creutzfeldt-Jakob disease (vCJD) agent and, theoretically, the Creutzfeldt-Jakob disease (CJD) agent. No cases of transmission of viral diseases, vCJD or CJD, have been associated with the use of COAGADEX.
In clinical studies, the most common adverse reactions (frequency ≥5% of subjects) with COAGADEX were infusion site erythema, infusion site pain, fatigue and back pain.
Please see complete Prescribing Information for COAGADEX.
You are encouraged to report negative side effects of prescription drugs to the FDA. Visit https://www.fda.gov/medwatch, or call 1-800-FDA-1088.
You may also call Kedrion at 1-866-398-0825 or email US_Medicalinfo@kedrion.com.